
In particular, examining the behavioral phenotypes of 14-3-3γ null mice is clinically important because 1) genetic mutations of ywhag gene have been recently associated in neurodevelopmental disorders and 2) the ywhag gene is located at 7q11.23 chromosomal abnormalities (deletion or duplication) at this locus are strongly linked to Williams-Beuren syndrome (WBS), which is presented with developmental delay, intellectual disabilities, and epilepsy. In addition, by directly interacting with ataxin-1 (spinocerebellar ataxia type 1), α-synuclein (Parkinson's disease), and TSC2 (tuberous sclerosis complex 2), 14-3-3γ is deeply involved in these neuropsychiatric diseases. ġ4-3-3γ, encoded by ywhag gene, is one of seven isoforms (β, γ, ε, σ, ζ, τ, and η) and its increased expression has been reported in Creutzfeldt-Jakob disease and ischemia. 14-3-3ζ KO mice displayed hyperactive behaviors and cognitive deficits, as well as abnormal neuronal migration in the hippocampus during development. In these studies, 14-3-3ε knockout (KO) mice displayed enhanced anxiety-like behaviors and memory deficits as well as morphological abnormalities in the hippocampus.

The behavioral phenotypes of two 14-3-3 isoform-specific null mice have been reported. These mice also display schizophrenia-like behaviors. In addition, transgenic mice expressing difopein, a 14-3-3 peptide inhibitor, displayed impairments of hippocampus-dependent learning and memory tasks, and long-term synaptic plasticity of hippocampal synapses. 14-3-3 proteins have also been shown to be involved in axon growth and pathfinding as well as neuronal regeneration. Since 14-3-3 proteins are enriched in the brain, they have been reported to be involved in a broad range of brain functions, and neurological and psychiatric diseases. The interaction of 14-3-3 proteins with specific partners affects their stability, localization, and activities within the cell. 14-3-3 proteins act as a dimer by binding to phosphorylated target proteins at specific site(s), causing a conformational change. In addition, 14-3-3γ heterozygote mice may be a potential model to study the molecular pathophysiology of neuropsychiatric symptoms.ġ4-3-3 proteins are a family of ubiquitously expressed adaptor proteins that are involved in diverse cellular processes, such as intracellular signaling, cell-cycle control, apoptosis, neuronal migration, and protein trafficking by regulating hundreds of different “client” proteins. These results suggest that 14-3-3γ levels may be involved in the developmental manifestation of related neuropsychiatric diseases. In addition, in behavioral analyses, we found that 14-3-3γ heterozygote mice display hyperactive and depressive-like behavior along with more sensitive responses to acute stress than littermate control mice.
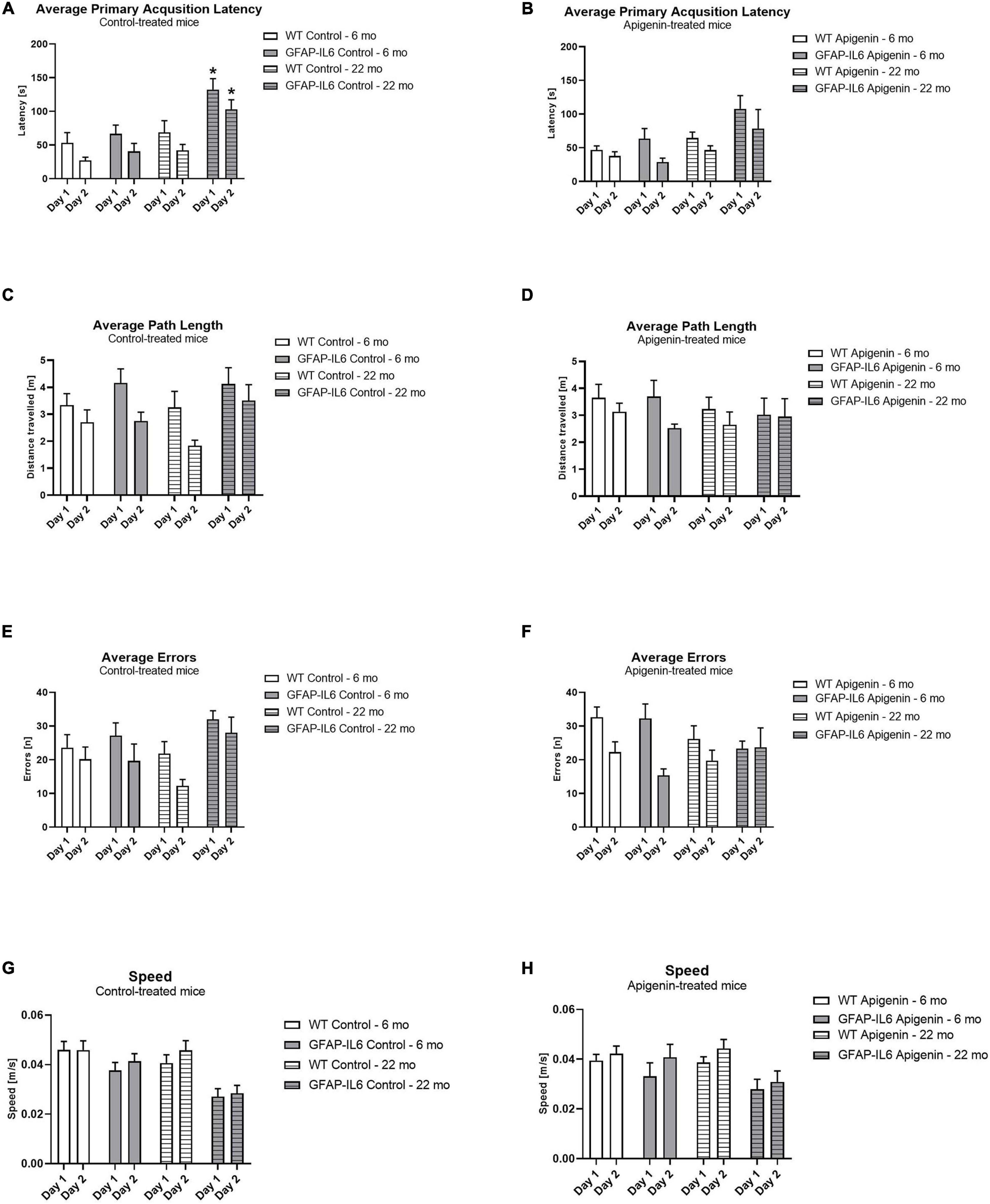
Here, by using 14-3-3γ deficient mice, we found that homozygous knockout mice were prenatally lethal, and heterozygous mice showed developmental delay relative to wild-type littermate mice. However, behavioral abnormalities related to 14-3-3γ deficiency are largely unknown. Williams-Beuren syndrome and Creutzfeldt-Jakob disease). Especially in the brain where 14-3-3γ is enriched, it has been reported to be involved in neurological and psychiatric diseases (e.g. 14-3-3γ plays diverse roles in different aspects of cellular processes.
